Body
After suffering a stroke, patients often are unable to use the arm on their affected side. Sometimes, they end up holding it close to their body, with the elbow flexed.
In a new study published in the Proceedings of the National Academy of Arts and Sciences, Shirley Ryan AbilityLab researchers — including study senior author Wendy Murray, PhD, research scientist; study first author Amy Adkins, a PhD student in Dr. Murray's lab; and Julius Dewald, research scientist and co-director, C-STAR Clinical Core — have discovered that, in an attempt to adapt to this impairment, muscles actually lose sarcomeres.
This is the first study ever to observe this phenomenon in humans.
Stacked end to end (in series) and side to side (in parallel), sarcomeres are muscles' smallest, most basic building blocks. They make up the length and width of muscle fibers. By imaging biceps muscles with three noninvasive methods, the researchers found that stroke patients had fewer sarcomeres along the length of the muscle fiber, resulting in a shorter overall muscle structure.
This finding is consistent with the common patient experience of abnormally tight, stiff muscles that resist stretching, and it suggests that changes in the muscle potentially amplify existing issues caused by stroke, which is a brain injury. The team hopes this discovery can help improve rehabilitation techniques to rebuild sarcomeres, ultimately helping to ease muscle tightening and shortening.
“This is the most direct evidence yet that chronic impairments, which place a muscle in a shortened position, are associated with the loss of serial sarcomeres in humans,” said Dr. Murray. “Understanding how muscles adapt following impairments is critical to designing more effective clinical interventions to mitigate such adaptations and to improve function following motor impairments.”
First Demonstration in Humans
Body
Measuring just 1.5 to 4.0 microns in length, sarcomeres comprise two main proteins: actin and myosin. When these proteins work together, they enable a muscle to contract and produce force. Although previous animal studies have found that muscles lose serial sarcomeres after a limb is immobilized in a cast, the phenomenon had never before been demonstrated in humans. In the animal studies, muscles that were shorter because they lost serial sarcomeres also became stiffer.
“There is a classic relationship between force and length,” said Amy Adkins. “Given that the whole muscle is composed of these building blocks, losing some of them affects how much force the muscle can generate.”
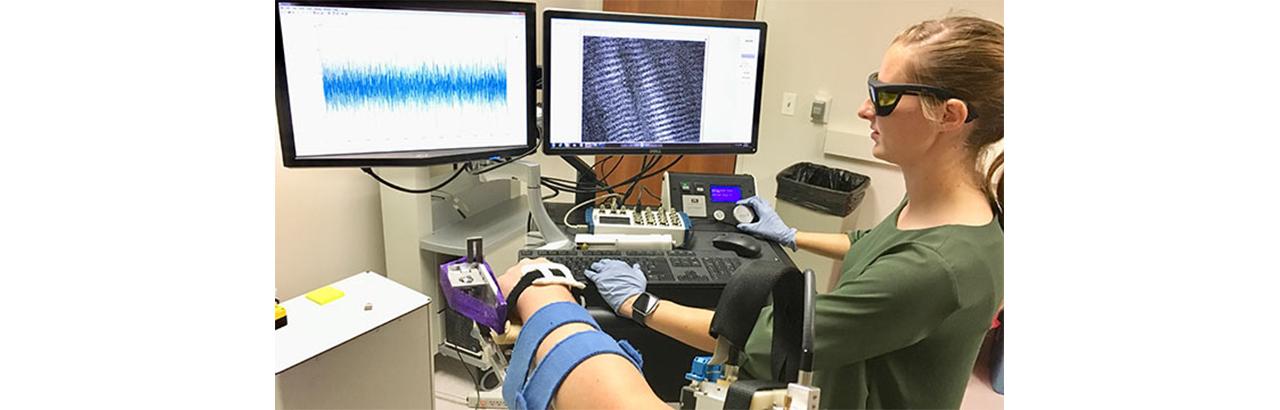
To conduct the study in humans, the researchers combined three non-invasive medical imaging techniques: MRI to measure muscle volume, ultrasound to measure bundles of muscle fibers and two-photon microendoscopy to measure the microscopic sarcomeres.
Imaging Opens New Possibilities
Body
Combining these technologies at Northwestern and Shirley Ryan AbilityLab, the researchers imaged biceps from seven stroke patients and four healthy participants. Because stroke patients are more affected on one side of their body, the researchers compared imaging from the patients’ affected side to their unaffected side as well as to images from the healthy participants.
The researchers found that the stroke patients’ affected biceps had less volume, shorter muscle fibers and comparable sarcomere lengths. After combining data across scales, they found that affected biceps had fewer sarcomeres in series compared to the unaffected biceps. The differences between stroke patients’ arms were greater than in healthy participants’ arms, indicating that the differences were associated with stroke.
By combining medical imaging to better view muscle structure, the study also establishes that it is possible to study muscle adaptations in sarcomere number in humans. Before two-photon microendoscopy, human studies were limited either to examining dissected tissues in anatomy labs, which give imperfect insight into how muscles adapt to injury and impairment, measuring sarcomere lengths during surgery or from a muscle biopsy, which restricts who can participate in the study.
“In almost every facet of our world, there is an important relationship between how something is put together (its structure) and how it works (its function),” the researchers said. “Part of the reason medical imaging is such a valuable resource and clinical tool is that this is also true for the human body, and imaging gives us an opportunity to measure structure.”
In addition to their roles at Shirley Ryan AbilityLab, Dr. Murray is a professor Biomedical Engineering at Northwestern University's McCormick School of Engineering and a professor of Physical Medicine and Rehabilitation at Northwestern University Feinberg School of Medicine; and Dr. Dewald is a professor of Physical Therapy and Human Movement Sciences and of Physical Medicine and Rehabilitation at Feinberg, and professor of Biomedical Engineering at McCormick.