Body
HIGHLIGHTS:
- Cullin-RING ligases play a pivotal role in proteins disposal and turnover in the cell (think of it like a "spring cleaning" occurring regularly inside the cell).
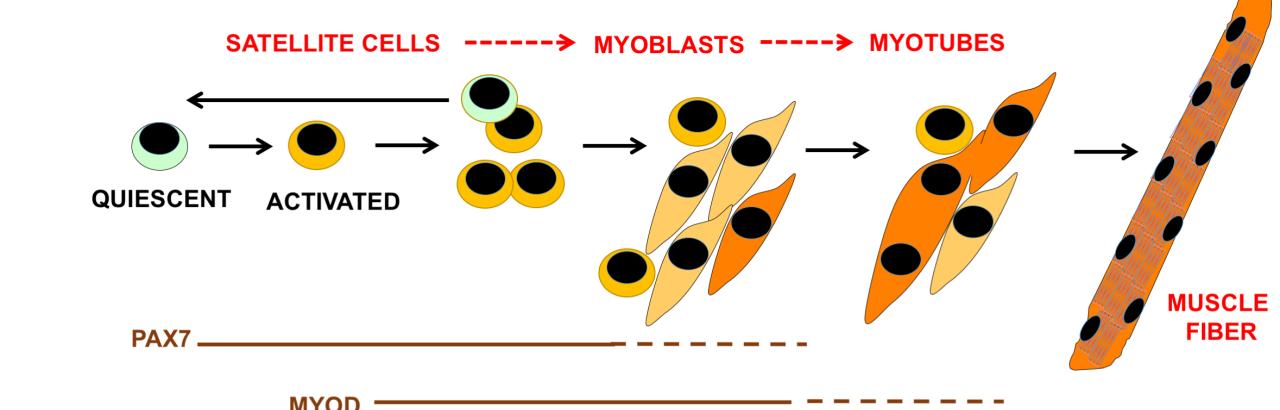
- They are essential for skeletal muscle fibers growth and differentiation.
- A cullin inhibitor, which is also used in cancer therapy, seems to be blocking muscle fiber formation in vitro.
- Cullin inhibition may have clinical implications for cancer patients in rehabilitation.
ABSTRACT:
"The role of cullin E3-ubiquitin ligases for muscle homeostasis is best known during muscle atrophy, as the cullin-1 substrate adaptor atrogin-1 counts among the most well-characterized muscle atrogins. We investigated, whether cullin activity was also crucial during terminal myoblast differentiation and aggregation of acetylcholine receptors for the establishment of neuromuscular junctions in vitro.
The activity of cullin E3-ligases is modulated through posttranslational modification with the small ubiquitin-like modifier nedd8. Using either the Nae1 inhibitor MLN4924 (Pevonedistat) or siRNA against nedd8 in early or late stages of differentiation on C2C12 myoblasts, and primary satellite cells from mouse and human, we show that cullin E3-ligase activity is necessary for each step of the muscle cell differentiation program in vitro. We further investigate known transcriptional repressors for terminal muscle differentiation ZBTB38, Bhlhe41 and Id1. Due to their identified roles for terminal muscle differentiation, we hypothesize that accumulation of these potential cullin E3-ligase substrates may be partially responsible for the observed phenotype.
MLN4924 is currently undergoing clinical trials in cancer patients, and our experiments highlight concerns on the homeostasis and regenerative capacity of muscles in these patients who often experience cachexia."
Mentioned Profile