Body
Targeted Muscle Reinnervation (TMSKR) is a pioneering surgical procedure that provides easier, more intuitive prosthesis control for individuals with arm amputation. It was developed by Todd A. Kuiken, MD, PhD, of the Shirley Ryan AbilityLab, and Gregory A. Dumanian, MD, of Northwestern Memorial Hospital.
During TMSKR, nerves in an amputee’s residual arm are transferred to target muscles that no longer perform a useful function because of the amputation. These nerves, which previously controlled the elbow, wrist, and hand of the missing arm, grow into (or reinnervate) the target muscle. The target muscles act as biological amplifiers of control signals from these nerves, generating electrical signals that can be used to control a prosthesis.
TMSKR makes prosthesis control more intuitive by creating additional control sites, and control signals intended for the missing elbow, wrist, or hand can be used to control those same movements in the prosthesis.
TMSKR is now a standard surgical procedure and has been performed at medical centers nationwide, including the San Antonio Military Medical Center and Walter Reed National Military Medical Center.
Our book, Targeted Muscle Reinnervation: A Neural Interface for Artificial Limbs (CRC Press, 2013), is a comprehensive guide to TMSKR, detailing the scientific basis for the procedure and the principles of prosthetic fitting and occupational therapy for TMSKR patients. The functional outcomes for the first people to undergo this procedure are described, and the possible role of TMSKR in the prevention and treatment of end-neuromas and in providing sensory feedback to prosthesis users is also discussed.
Introduction to Targeted Reinnervation
Targeted Muscle Reinnervation as a Strategy for Neuroma Prevention
Rehabilitation of the Targeted Muscle Reinnervation Patient
Occupational Therapy for the Targeted Muscle Reinnervation Patient
Introduction to Targeted Reinnervation
Body
An estimated 41,000 people in the United States live with amputations at or above the wrist. Many more people are affected by arm amputation throughout the world.
Loss of an arm causes a severe disability, affecting one's ability to perform daily tasks and often the emotional well-being of the individual.
The most common reason for arm amputation is trauma, such as industrial accidents or combat-related injuries. Many individuals with upper-limb amputations are active, young adults who must spend decades living without their arm or hand.
Dysvascular disease and cancer are less common causes of arm loss; and although rare, congenital limb deficiency affects the upper limb more than 50% of the time.
The optimal treatment for arm loss is to use a prosthetic arm; however, the dexterity and functionality of the intact hand and arm set high expectations for arm prostheses, and pose the technological challenge of developing devices that are both functional and easy to control.
Scientific Basis
Body
The brachial plexus nerves carry all the motor control information needed to move the arms, hands, and fingers. This vast trove of control information becomes inaccessible after arm amputation. TMSKR provides access to this information by enabling severed arm nerves to reinnervate redundant target muscles, which serve as natural biological amplifiers of these neural control signals. TMSKR has been performed successfully as long as six years after amputation of an arm.
Two basic features of neuromuscular physiology underlie the concept of TMSKR:
(1) Studies in animals and people have shown that even after limb amputation, the brain continues to transmit control signals through the severed residual nerves. This continues for long time periods—perhaps indefinitely.
(2) Animal studies have shown that severed nerves can grow into (reinnervate) different (non-native) muscle, and that this muscle contracts in response to signals from the new nerve.
Basic Principles of TMSKR
Successful TMSKR depends on two key factors:
(1) Good reinnervation of target muscles.
(2) Generation of strong, independent EMG signals.
For successful target muscle reinnervation:
- In a rat reinnervation model, bigger nerves (with more
motor neurons) ensure better muscle recovery. - Large brachial plexus nerves (8-10 mm diameter) are transferred to much smaller (up to 1 mm) motor nerve branches. Such hyper-reinnervation makes it more likely that all available motor endplates are reinnervated, thus ensuring good muscle recovery.
- The target muscle is denervated prior to nerve transfer. This ensures that the target muscle only contracts in response to intended arm movement, and that the maximum number of motor endplates are available for reinnervation by the transferred nerve.
For strong, independent EMG signals:
- Separating adjacent target muscles creates more
independent EMG signals. - Separate target muscles to prevent crosstalk.
- Remove fat overlying the target muscle.
Surgical Techniques
Body
The goal of TMSKR surgery is to enhance an individual’s control of a myoelectric prosthesis.
Myoelectric devices are controlled by small electrical signals (EMG signals), which are generated by contraction of muscles in the residual limb. EMG signals are detected by electrodes placed on the skin surface.
EMG signals are generated in response to neural signals, sent from the brain, when the individual attempts to move their limb. These signals continue to be sent even after amputation of the limb; however, this valuable control information cannot be accessed.
TMSKR surgery involves transfer of severed brachial nerves to new target muscles – muscles that are biomechanically redundant because of the amputation.
After TMSKR, neural signals from transferred nerves cause target muscles to contract, generating new EMG control signals. TMSKR thus allows access to neural control information that was lost due to amputation.
TMSKR makes prosthesis control more intuitive because the transferred brachial nerves carry control signals intended for the missing limb: attempts to move the missing limb generate EMG signals that can be used to control corresponding movements in a prosthesis.
TMSKR surgery requires an understanding of the anatomy and functions controlled by the four major infraclavicular branches of the brachial plexus.
The four nerves involved in TMSKR surgery are the:
- Musculocutaneous nerve
- Radial nerve
- Median nerve
- Ulnar nerve
For more details about the surgical techniques of TMSKR, please view our training guide video for clinicians.
Targeted Muscle Reinnervation as a Strategy for Neuroma Prevention
Body
People with limb loss must often live with neuromas, benign yet painful growths of nerve tissue that develop at the end of severed nerves. Consisting of disorganized axons encased in scar and fibrous tissue, neuromas cause a focal pain that is often difficult to treat.
Neuroma pain is caused by: (1) mechanical or chemical irritation of axons in the neuroma and (2) spontaneous firing within the neuroma and retrograde axonal sprouts proximal to the neuroma.
People with neuromas often feel pain when they wear prostheses, which can put pressure on neuromas and create intense discomfort. As a result, neuroma pain can interfere with a person’s ability to comfortably wear and use a prosthesis.
Between 10 to 25% of individuals with major limb amputations will develop chronic localized pain as a result of symptomatic neuromas. Neuroma pain is reported in as many as 71% of individuals who have had a traumatic amputation.
Many strategies have been investigated to either stop neuroma formation or ease the resulting pain, including pharmacologic treatments, modification of the prosthesis, scar massage, surgery, and manipulation of the bone or soft tissues. Unfortunately, these treatments may be temporary and ineffective, as proximal axons inevitably form another neuroma.
TMSKR may prevent or reduce neuroma formation by providing a denervated muscle target for the transected nerves to grow into. TMSKR allows regenerating axons to grow into the target muscle instead of forming a neuroma. In other words, TMSKR gives the severed nerves somewhere to go and something to do.
Evidence
Preclinical Studies
- In a rabbit neuroma model, previously amputated nerve stumps were transferred to a denervated rectus abdominus flap, after excision of end neuromas. After 10 weeks, the histological morphology of transferred nerves resembled non-amputated nerve controls more closely than amputated nerves with neuromas, suggesting that TMSKR prevented reformation of end neuromas.
- In a rat hind limb model, mixed (sensory + motor) or sensory nerves were transferred to either: (A) the motor point of a denervated target muscle (analogous to a TMSKR procedure); (B) the motor point of an innervated target muscle; or (C) buried in muscle. After 16 weeks, histological and gross anatomical comparisons for both types of nerves indicated that transfer to the motor point of a denervated muscle (group A) significantly reduced neuroma formation.
Initial Clinical Evidence
- Following TMSKR procedures at Northwestern Memorial Hospital, five out of nine shoulder disarticulation patients who reported neuroma pain prior to their TMSKR procedure reported no neuroma pain after TMSKR.
- At the Harborview Medical Center at the University of Washington in Seattle, 20 patients reported feeling neuroma pain prior to TMSKR; however, after TMSKR surgery, none of these patients reported neuroma pain.
- In a retrospective study analyzing data from NMH and the San Antonio Military Medical Center, of 15 patients with neuroma pain before TMSKR, 14 experienced complete resolution of pain in the transferred nerves.
- In individuals who did report continued neuroma pain following TMSKR surgery, the source of the pain in 2 out of 3 individuals was the lateral antebrachial cutaneous nerve, which was not transferred to muscle during the TMSKR procedures. In the third patient, the neuroma pain was reduced, although not eliminated, by TMSKR surgery.
These results suggest that TMSKR may be a new surgical option to prevent neuroma formation or to treat existing neuromas.
Rehabilitation of the Targeted Muscle Reinnervation Patient
Body
TMSKR involves elective surgery followed by a long post-surgical reinnervation period, as well as a significant time commitment for prosthetic fitting and occupational therapy training. Successful outcomes depend on careful screening of potential candidates, together with a comprehensive, team-based approach to rehabilitation.
Prior to TMSKR surgery, patients should undergo a thorough medical exam, an assessment of emotional and mental health, and an evaluation of likely compliance with the requirements for successful rehabilitation.
Presurgical evaluation
Appropriate patient selection can determine the success or failure of TMSKR surgery. In order to be considered a candidate for TMSKR, patients must:
- Have a shoulder disarticulation or transhumeral amputation - TMSKR has only been performed on patients with these type of amputations; however, researchers are currently investigating how individuals with transradial and transfemoral amputations may benefit from TMSKR.
- Be willing to live without their prosthesis for 3 to 6 weeks after surgery until their soft tissues heal.
- Be motivated enough to follow a lengthy recovery and training process.
The health and viability of residual musculature and nerves should be assessed. Brachial plexopathy is a contraindication for TMSKR, as healthy nerves and muscles are necessary for robust reinnervation. In addition, the examination should evaluate:
- Proximal injury to shoulder complex—common in traumatic amputation.
- Impairments to the sound limb, which may be exacerbated due to overuse injuries.
- Core strength and posture.
- Soft tissue deficits, bony prominences, split-thickness skin grafts or scars, which may impede prosthesis wear, but may be surgically addressed during the TMSKR procedure.
- A cognitive and emotional assessment as many individuals with amputation struggle with emotional health issues, depression, or anxiety, which may affect success of the TMSKR procedure. Such issues must be monitored by the clinical team and addressed before surgery. Consultation with a psychologist is recommended.
- Traumatic brain injury (TBI) is possible following a traumatic amputation.
- If TBI is suspected, cognitive screening is recommended to ensure that the patient is able to follow directions and comply with treatment.
- TMSKR should be delayed to allow patients to recover from mild or moderate TBI.
Evaluation of likely compliance
- To maximize the likelihood of compliance, the patient must thoroughly understand the TMSKR procedure, the timeframes involved, and the rehabilitation process.
- The patient must understand what TMSKR will do for them: it will not make their prosthesis more comfortable or less heavy. Previous abandonment of a prosthesis for these reasons may suggest that improved control may not result in the patient using the device.
- A history of noncompliance with treatment may indicate lower likelihood of compliance with the TMSKR process.
Patient and team education
Patient and team education is vital so that informed medical decisions can be made to ensure the success of the procedure and that patients receive the full benefits of TMSKR.
The TMSKR surgery is only the initial part of a multi-step recovery process. Pre- and post-surgery, patients should be educated on the specific details of their TMSKR procedures, as well as expectations for recovery and any other potential complications.
In addition, all members of the rehabilitation team—surgeons, physiatrists, occupational therapists, prosthetists—must understand which nerve transfers will be performed during surgery so successful rehabilitation recommendations and timelines can be created.
After TMSKR, prosthetic fitting should be delayed pending complete reinnervation and achievement of adequate strength in reinnervated muscles.
Potential surgical complications
TMSKR surgery has the same risk of infection as any other soft tissue surgery. However, some potential complications specific to TMSKR include:
- Risk of target muscle paralysis.
- Neuroma pain (although evidence suggests that TMSKR may prevent or treat neuroma pain, any cut nerve has the potential to develop painful end-neuromas).
- Phantom limb pain
- Transfer sensation—if skin is denervated during the TMSKR procedure, sensory afferents from transferred nerves may reinnervate this skin. Touching the reinnervated skin may allow the patient to feel sensations, often described as a “tingling feeling,” which seem to come from their missing limb. Although patients should be aware of this possibility, most patients have not found this to be burdensome.
Prosthetic Fitting
Body
TMSKR surgery creates at least two additional control sites in transhumeral patients and up to four sites for shoulder disarticulation patients, depending on the number of nerve transfers. Proper prosthetic fitting –including component selection and socket design – is necessary so that users can experience the full benefits of TMSKR.
Although many standard prosthetic fitting techniques apply to those who undergo TMSKR, the prosthetic fitting of TMSKR patients poses some unique challenges. These include:
- Identifying optimal locations for additional control sites.
- Placing additional electrodes into well-fitting sockets.
- Accommodating increased soft tissue movement.
- Allowing additional time for strengthening contraction and isolation of signals from reinnervated muscles.
Successful fitting requires collaboration between an experienced prosthetist with a comprehensive understanding of myoelectric fittings, a physiatrist, an occupational therapist, and the surgical team.
Importantly, in addition to careful fitting of the prosthesis before and after TMSKR surgery, the device must be adapted to accommodate control changes during the lengthy reinnervation process.
Prior to TMSKR
The prosthetist and physician should evaluate a person’s residual limb length, nerve and muscle health, past experience using a myoelectric prosthesis, willingness and aptitude to learn new control techniques, and emotional health, among other factors.
For recent amputees, fitting with a traditional myoelectric or body-powered prosthesis prior to TMSKR surgery may help them adjust to the weight and function of a prosthesis. After TMSKR surgery, these patients can then focus solely on the new control techniques.
Pre-surgical fitting for patients who undergo TMSKR long after their initial amputation depends on their previous experience with a prosthetic device.
Interim Reinnervation Period
After surgery, as the limb is stabilizing, a prosthetist should begin discussing and considering with the patient:
- Prosthetic Fitting Considerations
- Control Strategies/Mode Selection (transhumeral vs. shoulder disarticulation)
- Socket Fitting (tolerance to force distribution, active/passive use, suspension)
- Post-surgical education and training
During this interim period, it may also be necessary to:
(1) Alter electrode locations to accommodate any muscle body shifts that occurred during surgery.
(2) Increase electrode gains to accommodate the decrease in active muscle mass following surgery as a result of the denervation of some muscle segments.
After Reinnervation
Control site selection should be delayed until reinnervation begins to stabilize around 6 months after surgery.
During this time, an occupational therapist may provide training to help the person increase their muscle endurance to strengthen EMG signals.
Once reinnervation has stabilized, myotesting, prosthetic fitting, component selection, and training can begin. Ideally, before myotesting begins, the prosthetist and surgeon should examine the patient to review the nerve transfers performed.
Occupational Therapy for the Targeted Muscle Reinnervation Patient
Body
Effective occupational therapy after TMSKR surgery ensures that patients gain intuitive control of their prostheses and receive the full benefits of TMSKR. During pre-operative and post-operative training sessions, therapists must consider issues unique to TMSKR patients and plan accordingly. Following fat removal and soft tissue revision during TMSKR surgery, the size and shape of a patient’s residual limb may be altered, resulting in the need for socket modification or replacement. Any alteration of control methods, socket or electrode locations will require the occupational therapist to train the patient in using the prosthesis as intended. Knowledge of the location of each brachial plexus nerve transfer and the functions of each nerve is therefore essential for developing optimal control strategies.
Pre-surgery
Thoroughly discuss with the patient expectations and the post-operative program. Provide them with written and visual information.
Immediately After Surgery
All initial efforts after surgery should be focused on wound healing and controlling any surgical pain. Usually, it takes about 2–3 weeks for the surgical incision to heal and for the soft tissues to consolidate.
3-4 Weeks After Surgery
After the surgical incisions have healed, patients should begin an “exercise” program to strengthen relevant muscles for myoelectric prosthesis control. Individuals can first begin with mental practice—the repetitive motor imagery of a given movement. This mental practice generates changes in brain activation, improves motor performance, and can increase muscular force without direct muscle activation. Individuals should attempt to move each segment of the missing limb (fingertips, fingers, wrist, etc.) several times daily. They can do this by thinking about past activities they performed with their hands or arms, such as buttoning a shirt, making a sandwich, or playing an instrument. Initially there will be no visible response to this effort, as reinnervation has not yet occurred.
6-15 Weeks After Surgery
When specific muscle twitching occurs in response to efforts to move limb segments, patients should begin practicing gross motor movement patterns. The objective is to strengthen the targeted muscles by having the patient perform gross movement patterns using maximal contractions. Patterns that incorporate the distribution of the nerve transfers should be used, and movements should be performed bilaterally to further promote motor recruitment. Strengthening is achieved by progressively increasing the force and duration of muscle contractions, so exercise sets should be repeated at least four times per day. Early in training, the reinnervated muscle will still be weak and temporarily paralyzed, so muscles may tire quickly and patients will need to take frequent rest breaks.
The elbow should be excluded from all patterned exercises, as the coupling of elbow-flexion with hand-close, and elbow extension with hand-open can prevent successful TMSKR prosthetic fitting.
10-20 Weeks After Surgery
When consistent muscle target activation is palpable by the occupational therapist during gross movement exercises, patients should begin discrete movement strengthening. With discrete actions, the goal is to develop central actions that are strong, independent and isolated in preparation for fitting the new targeted reinnervation-control prosthesis.
24+ Weeks After Surgery
When myo-testing indicates three or more potential independent isolated myoelectric sites, prosthesis control training can begin. In particular, muscle relaxation techniques, which help patients avoid overexertion and fatigue, allow EMG signals to fall below a certain threshold so they can be used as an “off” signal.
Assessments and Education before Surgery
Before TMSKR surgery, physiatrists and therapists should particularly note a patient’s proximal shoulder on the injured side, as other joint injuries commonly coincide with traumatic arm amputations. Additionally, they should focus on a patient’s posture and core condition, which can affect prosthesis control, and:
- Make sure patients are strong and capable as possible using their current prosthesis.
- Assist patients in understanding the TMSKR concept.
- Prepare patients for the healing and recovery process, and foster realistic expectations and goals.
- Educate patients on techniques needed for increasing independence in basic self-care without a prosthesis.
In order to develop a unique occupational therapy plan, several outcome measures should be used to document changes in patients’ abilities to control prostheses, including the Canadian Occupational Performance Measure (COPM) and Patient Specific Functional Scale (PSFS).
Targeted Sensory Reinnervation
Body
Amputation results in not only the loss of the limb, but also loss of the receptors and pathways of the somatosensory system, which transmits sensory and positional information back to the brain.
Amputees must therefore rely exclusively on visual feedback to monitor their prostheses. This creates an overwhelming cognitive load for individuals with amputations: they must constantly watch their prosthesis to monitor where it is and what it is doing.
Tactile feedback is important for successful manipulation of objects and plays a role in learning of new tasks and refining active movement. Loss of sensory feedback also impairs the ability to perform fine motor tasks or to control limb posture, and affects the sense of ‘ownership’ of a limb.
TMSKR surgery was developed to allow access to motor control information intended for the missing limb, but an unexpected discovery after the first TMSKR procedure indicates that TMSKR may also provide a pathway to allow sensory feedback from a prosthesis.
Sensory nerve fibers from transferred nerves can grow through the muscle and into the overlying skin, reestablishing functional connections with sensory end organs. This phenomenon, termed transfer sensation, allows patients to “feel” as if their missing hand or arm is being touched. The video shows the reaction of an individual wearing a tactor when she ‘feels’ her missing arm after being touched on her reinnervated skin.
Transfer sensation differs from phantom limb sensation, which likely results from changes in functional connectivity and reorganization within the central nervous system (and thus is difficult to localize). Transfer sensation arises from the reactivation of amputated and transferred sensory afferents, and is thus felt as being precisely located to discrete areas on the missing limb.
A recent case study in which sensory nerve fascicles were directly transferred to surgically bisected cutaneous sensory nerves (during conventional TMSKR surgery) demonstrated discrete, separate sensory percepts of individual digits, which enabled precise sensory feedback while controlling a myoelectric arm.
Surgical Outcomes
Body
TMSKR is now an established procedure that has been performed on more than 100 patients in hospitals worldwide, including the San Antonio Military Medical Center (SAMMC), Walter Reed National Military Medical Center, and Northwestern Memorial Hospital (NMH) in Chicago.
The following data are based on surgical outcomes from TMSKR procedures performed from 2002 to 2012 at NMH and SAMMC. All data were obtained under approved protocols from the Institutional Review Boards of the appropriate institutions.
Patient Population
27 patients underwent TMSKR at NMH or SAMMC (17 subjects with transhumeral amputations; 10 subjects with shoulder disarticulation amputations).
Subjects ages ranged from 18-55, and average duration between amputation and TMSKR surgery was 16 months, with a range of 4 months to 6 years. Neither age nor time since amputation had any noticeable effect on outcomes.
All of these subjects had TMSKR performed on one side, although several patients had bilateral amputations.
Since publication of Targeted Muscle Reinnervation: A Neural Interface for Artificial Limbs, bilateral TMSKR has been performed on a patient with a right-side transhumeral amputation and a left-side shoulder disarticulation amputation.
Acute Surgical Outcomes
Mean operative time for transhumeral surgeries was 3 hours, 22 minutes; for shoulder disarticulation surgeries, the mean duration was 5 hours, 37 minutes. Surgical time was most likely longer for shoulder disarticulation surgeries because of the challenges imposed by altered anatomy due to high-impact injuries.
- 90% of patients were in the hospital for one night following surgery.
- 3 out of the 27 patients experienced delayed healing of the surgical incision; however, all patients went on to heal without the need for revision procedures.
- Mean duration of follow-up was 28 months.
In all, a total of 79 nerve transfers were performed, yielding robust EMG signals in 75 target muscle – a 95% success rate. However, not all signals were suitable for prosthesis control due to cross talk or difficulty in maintaining adequate electrode contact with skin.
Of the 27 patients who underwent TMSKR at NMH/RIC and SAMMC, 25 (93%) were subsequently fit with TMSKR-controlled myoelectric devices.
Postsurgical Pain Outcomes
- 15 of 19 patients who presented with phantom limb pain experienced an exacerbation of phantom limb pain symptoms following the procedure; however, pain returned back to pre-surgery levels 4-6 weeks later.
- Of 14 patients with neuroma pain, neuroma pain persisted in only 2 patients following TMSKR surgery.
Functional Outcomes
Body
We used three objective outcome measures to evaluate the success of TMSKR in six individuals:
1. Box and Block test – Subjects move one-inch blocks from one side of a box, over a partition, to the other side of the box. A higher score indicates improvement.
After TMSKR, individuals could move an average of 323% more blocks in the given time than they could before TMSKR.
2. Clothespin Relocation test – Subjects are timed as they move three clothespins from a horizontal bar, rotate them, and place them on a vertical bar. Adapted from the Rolyan Graded Pinch Exerciser, this test requires control of all three available degrees of freedom of the prosthesis, including wrist rotation. A lower score indicates improvement.
After TMSKR, subjects could manipulate and relocate clothespins on average of 49% more quickly than they could before TMSKR.
3. Assessment of Motor and Process Skills (AMPS) – Subjects choose from a list of familiar, relevant activities of daily living, such as making a sandwich.
This test evaluates motor skills, in particular how effectively and efficiently the subjects moves when grasping and manipulating objects during the task. The process skills evaluates the subject for task organization, planning, problem solving. Similar to the Box and Block test, a higher score indicates improvement.
After TMSKR, most subjects showed increased scores in both motor and process skills, and four out of five subjects demonstrated significant improvements in motor scores.
In addition, we used the Disabilities of the Arm, Shoulder, and Hand (DASH) outcome measure to evaluate subjective responses to TMSKR. The DASH consists of 30 scored questions on physical function, symptoms, and social functioning; the higher the score, the higher the level of disability experienced. Of the three subjects tested, all scored lower on the DASH after TMSKR.
Future Directions
Body
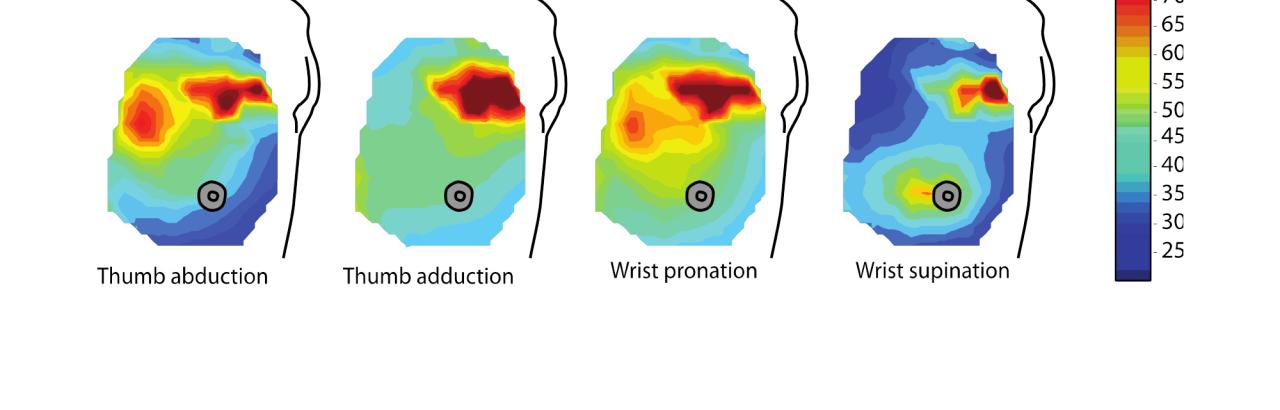
TMSKR & Pattern Recognition
Conventional EMG control techniques are based on the magnitude of EMG signals and give users basic control of powered prostheses. However, these techniques are relatively primitive; they use only a fraction of the available control information and are confounded by muscle cross talk—EMG signals from other adjacent muscles.
Consequently, these techniques require specific electrode placement, achieved through trial and error, to optimize control. This can be challenging in individuals with high level amputations, as they need to control several degrees of freedom but have few suitable control sites available.
In addition, because few residual muscles remain after above-elbow amputation, the user must often control several degrees of freedom using one muscle pair. This means that the user must use unintuitive contractions to control the prosthesis, for example, using the biceps and triceps to control the hand and wrist. The user must also switch the prosthesis, for example, from hand to elbow mode. Mode-switching is often done by co-contraction.
Pattern recognition provides an exciting new way to control a myoelectric prosthesis. Different attempted movements generate distinct, characteristic patterns of muscle activation, which in turn, generates unique EMG patterns—like an electrical fingerprint.
Computer algorithms can learn to recognize and distinguish these patterns; once trained, the algorithm can then decipher what the user intends to do and command the prosthesis to perform that movement.
Pattern recognition makes control of a prosthesis more intuitive, or natural: the user simply has to try to make the desired movement with their residual limb, and the prosthesis responds with the correct movement.
In addition, users do not have to switch the prosthesis between modes to control different degrees of freedom, so control is seamless.
Pattern recognition technology also does not require specific electrode placement, which makes clinical fitting simpler and faster.
Although pattern recognition works for people who have not had TMSKR, an individual who has undergone TMSKR may benefit even more from pattern recognition, because TMSKR creates additional EMG signals that can be incorporated into the patterns, creating a more detailed pattern. TMSKR also allows access to the rich neural information that is carried by transferred brachial plexus nerves for control of the arm, hand, and digits.
Recently, three TMSKR patients were fitted with a commercially available pattern recognition system through Coapt. One of these individuals had shoulder disarticulation amputation, and one had transhumeral amputation. The third individual had bilateral amputations (left shoulder disarticulation, right transhumeral), and was the first person to undergo bilateral TMSKR.
Coapt was launched in 2012 and has a technology transfer and license agreement with the Rehabilitation Institute of Chicago for the development of certain control technologies. Several current and former members of the Center for Bionic Medicine have management and ownership interests in Coapt.
Combining TMSKR with pattern recognition for other amputee populations
So far, TMSKR has been performed primarily on individuals with transhumeral or shoulder disarticulation amputations. However, as research progresses, TMSKR may become available for individuals with transradial and transfemoral amputations—dramatically increasing the number of individuals who can benefit from TMSKR.
Body
Development of this website was supported by the National Library of Medicine of the National Institutes of Health, Award Number G13LM011221. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.